Managing IBD therapy during pregnancy demands a multidisciplinary approach
Filip Marek1, Radoslav Hrivnák1, Ivo Rovný1, Petr Jabandžiev2,3, Karolína Poredská4, Vladimír Procházka Orcid.org 1, Dominika Ivanecká1, Vít Weinberger5, Petr Janků5, Lumír Kunovský1,4, Zdeněk Kala Orcid.org 1, Jiří Dolina Orcid.org 6, Daniel Bartušek Orcid.org 7
+ Affiliation
Summary
Crohn’s disease and ulcerative colitis are both chronic inflammatory bowel diseases (IBD). This article summarizes current best practice in treating pregnant patients with IBD, ranging from conservative therapy to endoscopy and imaging methods, including a description of surgical therapy indications. Female patients with IBD should ideally plan their pregnancies for when their disease is in remission. Patients in remission may also have complications during pregnancy, however the risk of complications is lower than in patients with active disease. Any chronic medications they were on before becoming pregnant (with the exception of teratogenic methotrexate) should remain unchanged. According to the current literature, pregnancy does not itself complicate the course of IBD. In cases of a severe relapse or an occurrence of complications in these patients, careful multidisciplinary cooperation is required, especially between the gastroenterologist, surgeon, radiologist, and gynaecologist. Surgical treatment is required only in cases of acute complications of IBD (such as acute severe colitis resistant to medical therapy, perianal abscess, and complications of IBD in the sense of such acute abdomen events as perforations, ileus due to a stenosis, or massive haemorrhage).
Keywords
Crohn’s disease, ulcerative colitis, inflammatory bowel disease, gravidity, surgery, endoscopy, porod, relaps
Introduction
Crohn’s disease (CD) and ulcerative colitis (UC), collectively termed inflammatory bowel diseases (IBD), are chronic bowel conditions most commonly diagnosed in patients between 20 and 40 years of age (i.e. in the patients’ fertile years). It is therefore important to discuss family planning with these patients and the possible risks associated with their treatment.
Fertility in both women and men with a controlled disease is generally comparable to that of the healthy population. In some individuals, however, this can be reduced, for example, by smoking, malnutrition, disease activity, as well as after pelvic surgery or proctocolectomy followed by reconstruction with ileopouch anal anastomosis (IPAA) [1,2].
Operations in the area of the lesser pelvis can lead to the formation of adhesions with a tendency to obstruction of the fallopian tubes and thus inability to conceive. In these patients, it is then necessary to consider another method of fertilization, such as in vitro fertilization (IVF). According to a recent study, women with IBD who underwent IVF achieved the same percentage of uncomplicated births as did women without this diagnosis [3]. Studies in women after IPAA, which showed the same proportion of completed healthy pregnancies after IVF, were similarly positive as in women without surgical therapy and in women without UC [4].
Lesser pelvic surgery can also affect male fertility. Due to the disruption of the autonomic nerves, anatomical changes and subsequent fibrosis in the lesser pelvis, it can lead to sexual dysfunction. In men, some drugs also have a negative effect on fertility. In particular, sulfasalazine and methotrexate may produce potential reversible disorders of spermatogenesis. In addition, methotrexate may also cause erectile dysfunction [5,6].
In the case of pregnancy, according to the European Crohn’s and Colitis Organisation (ECCO), the most important factor influencing possible complications is disease activity, in that this can increase the risks of premature birth, lower birth weights, and the need for caesarean section [1,7,8]. If a woman becomes pregnant during remission, the risk of IBD relapse during pregnancy is the same as in non-pregnant patients with IBD. If a patient becomes pregnant during a period of IBD activity, however, she is more likely to have both persistent activity and a risky course of the pregnancy itself [1]. Pregnancy is also likely to affect the course of IBD. According to a smaller retrospective study, quality of life improved in up to 50% of patients with IBD during pregnancy, and this may be related to the effects of pregnancy on the immune system [9,10]. After delivery, the risk of relapse does not increase in patients with CD. In cases of patients with UC, on the other hand, Pederson et al mention that the risk of postpartum relapse can be as much as 6-times greater [1,11]. Patients with UC are likely to have a higher risk of relapse during pregnancy (even if they are in remission during the conception period) compared to pregnant patients with CD or non-pregnant UC controls [12].
Medical therapy
Most medications used to treat IBD (except methotrexate, which is contraindicated) are considered safe during pregnancy. It is nevertheless important to educate patients before pregnancy about the nature of the drugs used and, if necessary, to dispel their fears of a possible negative effect on pregnancy. At the same time, patients need to be carefully instructed as to the risk of discontinuing established therapy, which in pregnancy may lead to a relapse of IBD and thereby endanger both mother and foetus [1,13].
Aminosalicylates and sulfasalazine
The use of aminosalicylates and sulfasalazine is safe during pregnancy; studies have shown no increased risk of miscarriage, ectopic pregnancy, or any teratogenic effect of this medication [1]. Sulfasalazine does interfere with folate absorption, however, and therefore folic acid is recommended to be administered to pregnant mothers at a dose of 2mg / day to prevent neurological malformations in neonates [1,14].
Antibiotics
The most commonly used antibiotics in patients with IBD include metronidazole, ciprofloxacin, and rifaximin. Although it is to be assumed that administration of metronidazole in the first trimester of pregnancy may cause cleft lip and palate in neonates, a recent study in 922 patients taking metronidazole during pregnancy did not confirm this assumption. Nevertheless, ECCO recommends that metronidazole should not be used in patients during the first trimester [1,15].
Additionally, ciprofloxacin may cause arthropathy in neonates due to its adverse effects on bone and cartilage. Therefore, ciprofloxacin also should be avoided in patients during the first trimester [1]. However, recommendations regarding the use of metronidazole and ciprofloxacin in the 1st trimester are not uniform and some associations consider them safe even in the 1st trimester as for example the American Gastroenterological Association (AGA) [16].
The effect of rifaximin on the course of pregnancy has not been adequately studied in patients with IBD. Therefore its administration is recommended only after an individual risk-benefit assessment [2].
Corticosteroids
Corticosteroids (GCS – glucocorticosteroids) are considered a safe and effective treatment for IBD relapse in pregnancy. Although they can cross the placenta, GCS are rapidly metabolized to less active metabolites and their concentration in foetal blood is very low [1,2]. According to some older studies, GCS therapy may increase the risk of orofacial malformations in the foetus during the first trimester [17]. Nevertheless, recent studies have not confirmed this statement. Moreover, GCS may increase the risk of hypertension, gestational diabetes, or preeclampsia in pregnant women. However, these risks have not been specifically confirmed in patients with IBD [1,2], though according to a study published by Leung et al [18] the effect of GCS on gestational diabetes formation was noted.
Although not much data on topical use of GCS exists, this also is considered a safe therapy having no adverse effects on pregnancy [1,19].
Thiopurines (azathioprine, 6-mercaptopurine)
Thiopurines are immunosuppressants used in patients with IBD to maintain surgically or drug-induced remission. Azathioprine and its metabolite 6-mercaptopurine cross the placental barrier. They can be detected in foetal erythrocytes and cause anaemia. Studies looking at the possible side effects of azathioprine are inconsistent in their conclusions. Some report possible immunological or haematological abnormalities in the foetus and the occurrence of premature births, while others regard azathioprine as safe [20,21]. As recommended by ECCO, azathioprine is considered safe and should not be discontinued or reduced during pregnancy due to the potential risk of IBD relapse [1,2]. However, it is not recommended to start azathioprine treatment during pregnancy due to the slow onset of action and the risk of side effects such as pancreatitis or possibly myelosuppression [6,16].
Cyclosporine and tacrolimus
Cyclosporine and tacrolimus are calcineurin inhibitors more commonly used in transplantology than in the treatment of IBD. Most of the work that deals with their safety in pregnancy is therefore based on data from cancer patients, according to which this therapy is safe and is not associated with developmental defects in newborns [1,22].
Methotrexate
Methotrexate (MTX – methotrexate) is one of the major teratogens and is therefore contraindicated during pregnancy. Although problem-free pregnancies have been documented, MTX can cause miscarriages, congenital malformations, growth retardation, or neurological malformations of the foetus. Therefore, termination of the pregnancy should be carefully considered if conception has taken place while taking MTX. Otherwise, one should discontinue MTX immediately and initiate folic acid substitution. With a view to pregnancy planning, it is recommended both for women and men (due to the long half-life of MTX degradation) to discontinue MTX 3–6 months before conception [1,23]. Studies in male patients treated with MTX during conception did not show an increased risk of fetal malformations or other complications [24,25]. Although the safety of administering MTX to men in the periceptive period is rather controversial, most recommendations still state the need to discontinue MTX in male patients 3–6 months before the planned conception.
Biological treatment
Infliximab, adalimumab, golimumab
Biological therapies approved and used in the Czech Republic for the treatment of IBD mainly include monoclonal antibodies blocking tumour necrosis factor (TNF). These so-called anti-TNF preparations (infliximab, adalimumab, golimumab) are considered safe during pregnancy on the basis of numerous studies [1,25–27]. According to the PIANO study, examining 1,000 pregnant patients on anti-TNF therapy, no higher risk of complications, including congenital malformations, was shown in mothers or newborns. However, children of mothers treated during pregnancy with anti-TNF drugs have been shown to be at increased risk of infections in the first year of life [28]. However, important data have been published recently by Duricova et al [29]. These data indicate that anti-TNF therapy administered to pregnant women did not have any negative impact on children’s postnatal development in terms of infectious complications, allergies, growth and psychomotor development when compared to children of unexposed mothers.
These monoclonal antibodies cross the placental barrier in the third trimester. Currently, there are inconsistent recommendations for treatment in the third trimester. The ECCO recommends temporarily discontinuing biological therapy at the end of the second trimester to reduce the effect of anti-TNF therapy on the foetus [1]. On the other hand, the AGA recommends that treatment should not be discontinued to minimize the risk of relapse [16]. Vaccination is contraindicated in the case of positive levels of biological drugs in the child‘s circulation and can be safely administered only after the drug has disappeared from the circulation (which can occur within the first 6 months after birth), due to the risk of infectious complications. If it is not possible to measure the levels in the umbilical cord blood/blood of the child, it is recommended to postpone this vaccination for at least 6 months, by when it is assumed that the levels will be negative for most children [1,30].
Indication for initiating biological therapy in pregnant patients with relapsed disease not responding to GCS therapy should be the same as in nonpregnant patients. Medical treatment of relapse should be similar to that in non-pregnant patients, except for initiation of treatment with thiopurines during pregnancy or treatment with methotrexate or tofacitinib [1,16,31].
Ustekinumab
Ustekinumab is a fully human monoclonal antibody that inhibits the activity of interleukin IL-12 and IL-23. Animal studies have shown no adverse effects of ustekinumab on pregnancy or on embryonal, foetal, or postnatal development [32]. The safety of ustekinumab in pregnancy is limited by the number of patients studied. Although the largest study showed that no difference was observed in congenital malformations or miscarriages compared to the healthy population [33]. The same recommendations for ustekinumab in pregnancy currently apply as for anti-TNF therapy.
Vedolizumab
This monoclonal antibody specifically binds to a4b7 integrin, which is preferentially expressed on helper T cells migrating to the gastrointestinal tract. Although there is not yet enough data on vedolizumab to clearly assess its safety during pregnancy (due to the low number of patients included in studies), it seems that vedolizumab does not have adverse effects on pregnancy or foetal development. A recent study in 24 pregnant patients, suggests that vedolizumab presents only low risk. Patients treated with vedolizumab during pregnancy did not show a higher number of abortions or a higher number of major or minor structural defects in the foetus compared to a healthy population of pregnant women [34]. In the largest study to date in 79 pregnant patients, no difference in the incidence of complications was observed compared to patients treated with anti-TNF or patients without anti-TNF or immunosuppressive therapy [35]. The same recommendations for vedolizumab also apply during pregnancy as for anti-TNF therapy.
Tofacitinib
Tofacitinib is a potent, selective inhibitor of the JAK (Janus kinase) family (primarily inhibiting JAK1, JAK2, JAK3, and, to a lesser extent, TYK2 – tyrosine kinase). Because tofacitinib has been shown to be teratogenic in rats and rabbits and adversely to affect both parturition and peri- and postnatal development, the use of tofacitinib during pregnancy is contraindicated as a precautionary measure. Women of childbearing potential should be instructed to use effective contraception during treatment with tofacitinib and for at least 4 weeks after the last dose in accordance with Czech State Institute for the Drug Control as published in Summary of Product Characteristics (SPC) [36]. Although according to the AGA, in the case of tofacitinib a washout period of 1 week should be adequate before attempting conception [16]. The safety of drugs used in pregnant IBD patients is summarized in Tab. 1 (modified according to van der Woude et al [1], Mahadevan et al [16], Magro et al [37] and Koželuhová et al [38]).
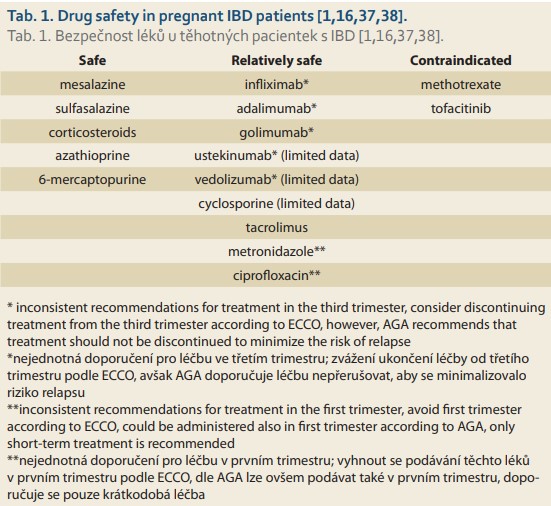
Endoscopic examination in pregnancy
According to ECCO guidelines, endoscopy (gastroscopy, sigmoidoscopy, colonoscopy) in pregnant patients with IBD is generally considered a safe diagnostic method. The data supporting this claim is nevertheless still insufficient. On the other hand, endoscopy is a safer option compared to radiological or surgical interventions. To reduce risks, endoscopy should be performed in pregnant patients only in cases (Fig. 1a, b) with clear clinical benefit, preferably in the second trimester of pregnancy. Furthermore, the cooperation of a gynaecologist/obstetrician and an anaesthesiologist is recommended during the examination [1,39].
The risks associated with performing endoscopic examinations in pregnant patients include, for example, the possibility of aspiration after gastroscopy due to incompetence of the lower oesophageal sphincter on the part of the mother and insufficient oxygenation and blood flow through the placenta on the part of the foetus. To prevent compression of the inferior vena cava and subsequent hypotension in pregnant women, endoscopies should be performed on the left side or prone position with the pelvis turned to the left, before, during, and after the examination [1,39].
Premedication before endoscopy must be chosen so that it is safe during pregnancy, and given in the lowest possible dose, but at the same time ensuring sufficient comfort of the patient during the examination. Of the commonly used premedications, fentanyl is considered safe in pregnancy, propofol is not recommended in the first and second trimesters, and probably the most commonly used benzodiazepines should not be given in the first trimester. Patients should be monitored during the examination (for saturation), and foetal heartbeat should be checked both before premedication and after the endoscopic procedure [1,39].
Use of imaging methods in IBD during pregnancy
Imaging methods comprise an important part of diagnostics in IBD. Their application comes mainly in diagnosis and monitoring but also in investigating acute conditions and complications of this serious disease. When examining pregnant women, however, the principle is that we must use imaging methods not involving exposure to radiation. This narrows the choice of modalities to ultrasonography (USG) and magnetic resonance imaging (MRI). Under certain, acute circumstances (e.g. toxic megacolon, ileus, and consideration of acute surgical treatment) and in the unavailability of MRI, it is possible to proceed with imaging methods using ionizing radiation. For these acute conditions, however, we select only those examinations that are essential for diagnosis of the pathological condition and follow the ALARA rule (as low as reasonably achievable) [40]. Obviously, the patient must be fully informed and give written consent.
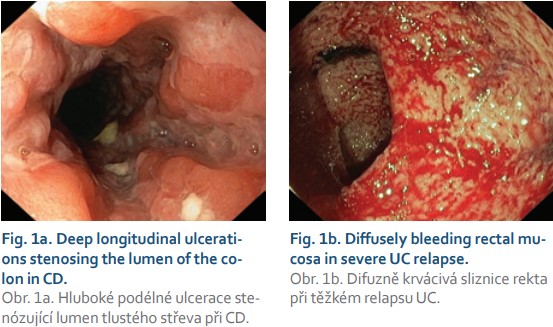
USG is currently the dominant imaging method in patients with IBD. Of course, this also applies to pregnant patients both in follow-up and in acute conditions. USG is considered a safe method in regard to possible side effects, and it is rapid and affordable. The method allows for overall assessment of the abdominal cavity, parenchymatous organs, and intestine. Depending on the stage of pregnancy, however, the assessment of the intestine may be limited. The course of the large intestine can be captured relatively well. Detection of the ileocecal region, terminal ileum, appendix, and small intestine can be problematic. However, if a sonographically affected section of the intestine is captured, we can decide whether this section is affected and whether there are signs of activity or possible complications in the environment (Fig. 2a, b). The basic attributes considered in an USG image in IBD include a reinforced intestinal wall, the nature of the involvement of individual layers of the intestinal wall, vascularization, the presence of enlarged lymph nodes, the length of an affected section, intestinal dilatation, free fluid, and the presence of fistulas or fluid collections [41].
MRI is a method with high sensitivity and specificity in evaluating changes in IBD. It is widely used in these patients and, together with transrectal sonography, is invaluable in the issue of anoperineal involvement. The method is able to evaluate the mural and extramural manifestations of the disease and to assess the extent of the activity. Compared to USG, its sensitivity is also higher in the detection of skip lesions. Other functions of MRI include the assessment of possible extraintestinal manifestations of IBD, such as primary sclerosing cholangitis, nephrolithiasis, or sacroileitis. Furthermore, MRI examination can serve as a non-invasive alternative for assessing IBD activity and therapeutic response.
MRI examination has generally wellknown contraindications, both absolute and relative. Examination of pregnant women is currently approached such that MRI in pregnant women in the second and third trimesters of pregnancy is generally considered safe [42]. In the first trimester, MRI is recommended and performed only with caution and its diagnostic benefit should be carefully considered [43]. Administration of contrast enhancers is contraindicated throughout pregnancy [44].
Of these imaging methods, USG is most often used in practice in pregnant patients with IBD. The use of MRI is possible, but in practice it is rather rare.
Childbirth methods in IBD patients
The decision whether birth should be conducted naturally or by caesarean section is typically discussed by a multidisciplinary commission, where the gynaecologist/obstetrician has the main say in the decision. Sometimes decisionmaking is influenced by complications in the underlying disease. While patients with UC have a percentage of caesarean delivery comparable to the general population, pregnant women with CD have a higher percentage [7].
IBD can affect the management of delivery in several ways. One of them is in preserving the function of the anal sphincter and associated continence in patients with perianal disease. Associated with this is the desire of mothers giving birth naturally to maintain good long-term continence in case of complications during vaginal delivery. There is also concern about the development of perianal disease after vaginal delivery. Although a smaller Canadian study showed only one case of developing perianal involvement after a complicated vaginal delivery [45], another larger study showed onset of perianal disease in as many as 18% of mothers out of 179 observed [46]. Conversely, patients with CD who do not have perianal involvement or whose perianal involvement is compensated or inactive during the birth may give birth vaginally, including episiotomy, because perianal complications are unlikely to occur within the underlying disease [47]. According to ECCO guidelines, caesarean section is recommended for pregnant women with active perineal involvement, as the risk of complications after vaginal delivery is significantly higher [48]. The main problem associated with this procedure is prolonged or complicated perineal healing in terms of inflammation, infections, and more frequent perineal rupture categories III and IV. In patients with CD after construction of ileorectal anastomosis, according to ECCO recommendations for IBD, the indication for caesarean section is specific to each case and the method of childbirth is chosen individually [1].
In patients with UC, vaginal delivery is recommended except for those who already have undergone proctocolectomy with IPAA [1,45]. Although vaginal delivery has been shown to be associated with pathophysiological changes in the anal sphincter, it is unclear whether this affects the long-term function of the pouch [49]. The physiology of the anorectum is often disturbed, as contraction pressures (squeeze pressure) are reduced and anorectal sonography shows additional defects of the anal sphincter, but these do not have a clinical effect on the function of the pouch [50]. This was shown in a large study of 232 women who gave birth vaginally after an already established IPAA and in whom functional dysfunction of the pouch was not demonstrated [51]. Similar results were published in a smaller study from Finland [52]. According to ECCO recommendations, in women with UC and IPAA (similar to patients with CD and ileorectal anastomosis), delivery by caesarean section is indicated relative to each case, with the approach being chosen individually [1]. Pregnant IBD patients with colostomy or ileostomy can deliver vaginally [48].
Surgical treatment
The indications for acute surgical treatment in pregnant patients with IBD do not differ from those in non-pregnant patients [1]. Acute exacerbation of IBD, unresponsive to medical treatment, is a greater risk to the foetus than is surgery itself [53]. Acute abdomen in all IBD patients and pregnant women in particular should be treated in specialized centres with 24-hour availability of a gastroenterologist, radiologist, and surgeon with experience in IBD surgery. In all patients with IBD, efforts are made to have the pregnancy planned for a period of disease remission. If an elective surgical solution is indicated for a patient, and especially for one with CD, it is appropriate to perform this operation before pregnancy. In these patients, we can safely perform laparoscopic surgery with anastomosis (mainly in the ileocecal region) without compromising pregnancy [54].
If a surgical solution is necessary for a pregnant IBD patient, we must consider not only the general recommendations but also the risks arising from pregnancy. In particular, it is necessary to consider surgical tactics based on the size of the foetus and uterus according to the progress of the pregnancy and to time the surgical procedure after consulting an erudite gynaecologist. Even in healthy women, surgery in the first trimester increases the risk of miscarriage, while in the third trimester, there is a risk of preterm birth due to technical difficulties in laparotomy [55,56]. If elective surgery is indicated during pregnancy, it should be scheduled between the 16th and 20th weeks when the foetus is securely implanted [57,58]. Depending on the progress of the pregnancy, both laparoscopic and open surgery can of course be considered. Laparoscopic operations are technically simpler and are recommended in the first and second trimesters of pregnancy for smaller uterine sizes [59]. Advantages of the laparoscopic approach include faster recovery, less trauma to the abdominal wall and lower postoperative morbidity. As pregnancy continues, laparoscopy becomes significantly more difficult. The laparoscopic approach to acute abdomen, though safe in pregnant women, has risks (miscarriages, premature births, low birth weight) comparable to those of open surgery [60–63].
The gynaecologist is to be involved in decision-making as to the management of surgical treatment, and especially in the period after 28 weeks of pregnancy when considering drug-related maturation of the foetus followed by a combined caesarean section and abdominal surgery [64,65]. Even this approach, however, will not allow us have a safe and clear operation in the area of the lesser pelvis, due to the size of the uterus, which will shrink to its original size within 4–6 weeks after birth.
Crohn’s disease
There is little data in the literature on the surgical treatment of pregnant patients with complicated CD. The largest such study includes results from just 15 patients [66]. For this reason, we must follow the results and procedures in nonpregnant patients with this disease. It is the duty of the dispensary gastroenterologist to consider surgical solutions in patients with stenosis of the terminal ileum in the period before conception to reduce the risk of developing an acute condition during pregnancy. Most patients with CD do not need surgery during pregnancy, but, in the event of a serious exacerbation or complication, we must always involve a multidisciplinary team Further, these patients should be treated in IBD treatment and surgery centres [67,68].
We most often indicate acute conditions such as obstruction unresponsive to medical treatment, bleeding, and perforation as reasons for surgical treatment [69–71]. We always undertake to pursue the most limited procedure. We can deal with the aforementioned conditions by resection of the small intestine, ileocolic resection, segmental resection, or anatomically defined resection of the colon.
In the case of risk factors for the healing of an anastomosis, we always choose a resection without an anastomosis with a stoma or only a stoma above the affected section. The foetus and the development of pregnancy would be significantly and negatively affected by any anastomotic complications or the development of a septic condition. Definitive resection is then to be planned electively in the months following childbirth [72].
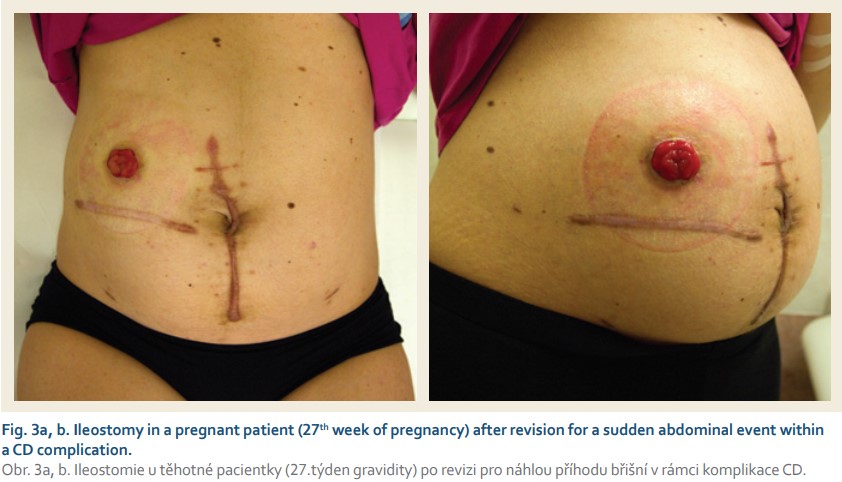
In case of need to perform a stoma, it is necessary to consider the placement. We must take into account the increasing volume of the uterus and thus prevent oppression of the stoma by the enlarged uterus with subsequent onset of ileus. Therefore, it is appropriate to perform a stoma more cranially than standard (Fig. 3a, b – stoma in a pregnant woman with CD) [73]. In case of subsequent ileostomy obstruction by the growing uterus, MRI examination of the abdomen can be performed to assess the aetiology of the ileus. As far as uterine compression is concerned, a conservative procedure is in place (e. g. catheter insertion into a stoma, and parenteral nutrition). Upon confirmation of any other mechanical cause (e.g. adhesion) revision of the abdominal cavity is indicated [74,75].
Perianal Crohn’s disease (PACD) is also related to CD. The treatment of perianal fistulas always involves a combination of medical and surgical therapy. That is in contrast to luminal involvement, where the treatment of milder forms is fully under the direction of a gastroenterologist. The aim of surgical treatment of PACD is primarily to control pelvic sepsis, improve quality of life, and maintain continence. Eradication of fistulas is only a secondary target. PACD treatment lasts for months to years [69]. Patients must always be examined anoscopically. If a more extensive examination, fistulotomy, drainage of the perianal abscess, or loose Seton placement is required, endoscopy under general anaesthesia is needed. In cases of extensive PACD, it is preferable to create a diverting stoma before pregnancy [76]. The final solution and eradication of the fistula then always follows at a later time. The PACD pregnant patient always has to consult the obstetrician to plan a caesarean section, which is fully recommended [48].
Ulcerative colitis
The standard procedure for UC, in the case of failure of conservative therapy (due to resistance, intolerance, or side effects) or risk of neoplasia, is to perform a proctocolectomy with IPAA [77,78]. During pregnancy, only acute surgical procedures are performed. In the case of UC, such are the development of acute severe colitis with toxic megacolon, massive bleeding, or perforation [70,79]. Rescue treatment of acute severe colitis must be conducted in close cooperation with a gastroenterologist and a gynaecologist. At the same time, frequent checking by a digestive surgeon is necessary. In pregnant patients, we approach the surgical solution earlier due to the risk of endangering the foetus with sepsis. The method of choice is a colectomy with a blind closure of the rectum stump and a terminal ileostomy [80]. With more advanced pregnancy and inaccessibility of the lesser pelvis and a high risk of bleeding from overcrowded pelvic venous plexuses, we are sometimes forced to leave a longer stump of the rectum as a mucosal fistula. It is always necessary to modify the location of the stoma according to the stage of pregnancy (discussed above). Manipulation of the uterus can cause premature birth, so, in some studies it is recommended to perform colectomy after the 28th week of pregnancy simultaneously with a caesarean section [64,65]. The final surgical solution is always planned for an adequate time after childbirth [80].
Conclusion
Managing the treatment of pregnant patients with IBD demands a multidisciplinary approach and should involve a gastroenterologist, surgeon, radiologist, and gynaecologist. Patients with IBD should be thoroughly informed before any planned pregnancy and reassured that, in most cases, the course of pregnancy is uncomplicated if the underlying disease is well handled. Established medication (except for teratogenic drugs explicitly) should be continued. Pregnancy does not worsen the course of IBD. A surgical solution is considered only for acute complications of IBD. The choice of delivery is at the direction of the gynaecologist and depends on the location of the IBD.
Abbreviations
ALARA – as low as reasonably achievable
AGA – American Gastroenterological Association
IPAA – ileo-pouch anal anastomosis
CD – Crohn’s disease
GCS – glucocorticosteroids
UC – ulcerative colitis
IBD – inflammatory bowel disease
ECCO – European Crohn’s and Colitis Organisation
MTX – methotrexate
PACD – perianal Crohn’s disease
TNF – tumour necrosis factor
USG – ultrasonography
MRI – magnetic resonance imaging
JAK – Janus kinase
TYK – tyrosine kinase
Submitted/Doručeno: 23. 1. 2021
Accepted/Přijato: 8. 3. 2021
Lumir Kunovsky, MD, PhD
Department of Surgery
University Hospital Brno
Faculty of Medicine Masaryk University
Jihlavska 20
625 00 Brno, Czech Republic
kunovsky.lumir@fnbrno.cz
To read this article in full, please register for free on this website.
Benefits for subscribers
Benefits for logged users
Literature
1. van der Woude CJ, Ardizzone S, Bengtson MB et al. The second European evidenced-based consensus on reproduction and pregnancy in inflammatory bowel disease. J Crohns Colitis 2015; 9(2): 107–124. doi: 10.1093/ecco-jcc/jju006.
2. Hashash JG, Kane S. Pregnancy and inflammatory bowel disease. Gastroenterol Hepatol (NY) 2015; 11(2): 96–102.
3. Oza SS, Pabby V, Dodge LE et al. In vitro fertilization in women with inflammatory bowel disease is as successful as in women from the general infertility population. Clin Gastroenterol Hepatol 2015; 13(9): 1641–1646. doi: 10.1016/j. cgh.2015.03.016.
4. Pabby V, Oza SS, Dodge LE et al. In vitro fertilization is successful in women with ulcerative colitis and ileal pouch anal anastomosis. Am J Gastroenterol 2015; 110(6): 792–797. doi: 10.1038/ajg.2014.400.
5. Shin T, Okada H. Infertility in men with inflammatory bowel disease. World J Gastrointest Pharmacol Ther 2016; 7(3): 361–369. doi: 10.4292/wjgpt.v7.i3.361.
6. Ďuricová D. Plánování těhotenství a léčba v graviditě. In: Lukáš M et al (eds). Idiopatické střevní záněty – Nové trendy a mezioborové souvislosti. Praha: Grada Publishing 2020.
7. Cornish J, Tan E, Teare J et al. A meta-analysis on the influence of inflammatory bowel disease on pregnancy. Gut 2007; 56(8): 830–837. doi: 10.1136/gut.2006.108324.
8. Bröms G, Granath F, Linder M et al. Complications from inflammatory bowel disease during pregnancy and delivery. Clin Gastroenterol Hepatol 2012; 10(11): 1246–1252. doi: 10.1016/j. cgh.2012.08.018.
9. Ananthakrishnan AN, Zadvornova Y, Naik AS et al. Impact of pregnancy on health-related quality of life of patients with inflammatory bowel disease. J Dig Dis 2012; 13(9): 472–477. doi: 10.1111/j.1751-2980.2012.00618.x.
10. Piccinni MP, Scaletti C, Maggi E et al. Role of hormone-controlled Th1-and Th2-type cytokines in successful pregnancy. J Neuroimmunol 2000; 109(1): 30–33. doi: 10.1016/s01 65-5728(00)00299-x.
11. Pedersen N, Bortoli A, Duricova D et al. The course of inflammatory bowel disease during pregnancy and postpartum: a prospective European ECCO‐EpiCom Study of 209 pregnant women. Aliment Pharmacol Ther 2013; 38(5): 501–512. doi: 10.1111/apt.12412.
12. de Lima-Karagiannis A, Zelinkova-Detkova Z, van der Woude CJ. The effects of active IBD during pregnancy in the era of novel IBD therapies Am J Gastroenterol 2016; 111(9): 1305–1312. doi: 10.1038/ajg.2016.254.
13. de Lima A, Zelinkova Z, Mulders AG et al. Preconception care reduces relapse of inflammatory bowel disease during pregnancy. Clin Gastroenterol Hepatol 2016; 14(9): 1285–1292. doi: 10.1016/j.cgh.2016.03.018.
14. Nielsen OH, Maxwell C, Hendel J. IBD medications during pregnancy and lactation. Nat Rev Gastroenterol Hepatol 2014; 11(2): 116–127. doi: 10.1038/nrgastro.2013.135.
15. Koss CA, Baras DC, Lane SD et al. Investigation of metronidazole use during pregnancy and adverse birth outcomes. Antimicrob Agents Chemother 2012; 56(9): 4800–4805. doi: 10.1128/AAC.06477-11.
16. Mahadevan U, Robinson C, Bernasko N et al. Inflammatory bowel disease in pregnancy clinical care pathway: a report from the American Gastroenterological Association IBD parenthood project working group. Gastroenterology 2019; 156(5): 1508–1524. doi: 10.1053/j. gastro.2018.12.022.
17. Carmichael SL, Shaw GM, Ma C et al. Maternal corticosteroid use and orofacial clefts. Am J Obstet Gynecol 2007; 197(6): 683–684. doi: 10.1016/j.ajog.2007.05.046.
18. Leung YP, Kaplan GG, Coward S et al. Intrapartum corticosteroid use significantly increases the risk of gestational diabetes in women with inflammatory bowel disease. J Crohns Colitis 2015; 9(3): 223–230. doi: 10.1093/ecco-jcc/jjv006.
19. Beaulieu DB, Ananthakrishnan AN, Issa M et al. Budesonide induction and maintenance therapy for Crohn’s disease during pregnancy. Inflamm Bowel Dis 2009; 15(1): 25–28. doi: 10.1002/ibd.20640.
20. Jharap B, de Boer NK, Stokkers P et al. Intrauterine exposure and pharmacology of conventional thiopurine therapy in pregnant patients with inflammatory bowel disease. Gut 2014; 63(3): 451–457. doi: 10.1136/gutjnl-2012-303615.
21. Cleary BJ, Källén B. Early pregnancy azathioprine use and pregnancy outcomes. Birth Defects Res A Clin Mol Teratol 2009; 85(7): 647–654. doi: 10.1002/bdra.20583.
22. Oz BB, Hackman R, Einarson T et al. Pregnancy outcome after cyclosporine therapy during pregnancy: a meta-analysis. Transplantation 2001; 71(8): 1051–1055. doi: 10.1097/00007890- 20010427 0-00006.
23. Kozlowski RD, Steinbrunner JV, MacKenzie AH et al. Outcome of first-trimester exposure to low-dose methotrexate in eight patients with rheumatic disease. Am J Med 1990; 88(6): 589–592. doi: 10.1016/0002-9343(90)90 522-f.
24. Grosen A, Kelsen J, Hvas CL et al. The influence of methotrexate treatment on male fertility and pregnancy outcome after paternal exposure. Inflamm Bowel Dis 2017; 23(4): 561–569. doi: 10.1097/MIB.0000000000001064.
25. Shannahan SE, Erlich JM, Peppercorn MA. Insights into the treatment of inflammatory bowel disease in pregnancy. Therap Adv Gastroenterol 2019; 12: 1756284819852231. doi: 10.1177/1756284819852231.
26. Schnitzler F, Fidder H, Ferrante M et al. Outcome of pregnancy in women with inflammatory bowel disease treated with antitumour necrosis factor therapy. Inflamm Bowel Dis 2011; 17(9): 1846–1854. doi: 10.1002/ibd.21583.
27. Restellini S, Biedermann L, Hruz P et al. Update on the management of inflammatory bowel disease during pregnancy and breastfeeding. Digestion 2020; 101(Suppl 1): 27–42. doi: 10.1159/000502886.
28. Mahadevan U, Martin CF, Sandler RS, et al. PIANO: a 1000 patient prospective registry of pregnancy outcomes in women with ibd exposed to immunomodulators and biologic therapy. Gastroenterology 2012; 142(5): S-149. doi: 10.1016/S0016-5085(12)60561-7.
29. Duricova D, Dvorakova E, Hradsky O et al. Safety of anti-TNF-alpha therapy during pregnancy on long-term outcome of exposed children: a controlled, multicenter observation. Inflamm Bowel Dis 2019; 25(4): 789–796. doi: 10.1093/ibd/izy294.
30. Zelinkova Z, van der Ent C, Bruin KF et al. Effects of discontinuing anti-tumour necrosis factor therapy during pregnancy on the course of inflammatory bowel disease and neonatal exposure. Clin Gastroenterol Hepatol 2013; 11(3): 318–321. doi: 10.1016/j.cgh.2012.10.024.
31. Aratari A, Margagnoni G, Koch M et al. Intentional infliximab use during pregnancy for severe steroid-refractory ulcerative colitis. J Crohns Colitis 2011; 5(3): 262. doi: 10.1016/ j.crohns.2011.02.004. 32. AlAmeel T, Al Sulais E. Ustekinumab and pregnancy: continue or cease before you conceive? Gastroenterology 2019; 157(5): 1435. doi: 10.1053/j.gastro.2019.05.077.
33. Geldhof A, Volger S, Lin CB et al. Pregnancy outcomes in women with psoriasis, psoriatic arthritis, Crohn’s disease and ulcerative colitis treated with ustekinumab. J Am Acad Dermatol 2020; 83(Suppl 6): AB74. doi: 10.1016/j. jaad.2020.06.382.
34. Bar-Gil Shitrit A, YaʼAcov AB, Livovsky DM et al. Exposure to vedolizumab in IBD pregnant women appears of low risk for mother and neonate: a first prospective comparison study. Am J Gastroenterol 2019; 114(7): 1172–1175. doi: 10.14309/ajg.0000000000000186.
35. Moens A, van der Woude CJ, Julsgaard M et al. Pregnancy outcomes in inflammatory bowel disease patients treated with vedolizumab, anti-TNF or conventional therapy: results of the European CONCEIVE study. Aliment Pharmacol Ther 2020; 51(1): 129–138. doi: 10.1111/apt.15539.
36. SÚKL. Xeljanz. 2021 [online]. Available from: https://www.sukl.cz/modules/medication/detail.php?kod=0238764.
37. Magro F, Gionchetti P, Eliakim R et al. ECCO. Third European evidence-based consensus on diagnosis and management of ulcerative colitis. Part 1: definitions, diagnosis, extra-intestinal manifestations, pregnancy, cancer surillance, surgery, and ileo-anal pouch disorders. J Crohns Colitis 2017; 11(6): 649–670. doi: 10.1093/ecco-jcc/jjx008.
38. Koželuhová J, Balihar K, Janská E et al. Terapie idiopatických střevních zánětů v graviditě. Interní Med 2015; 17(5): 218–222.
39. Maaser C, Sturm A, Vavricka RS et al. ECCO-ESGAR guideline for diagnostic assessment in IBD part 1: initial diagnosis, monitoring of known IBD, detection of complications. J Crohns Colitis 2019; 13(2): 144–164. doi: 10.1093/ecco-jcc/jjy113.
40. Uffmann M, Schaefer-Prokop C. Digital radiography: the balance between image quality and required radiation dose. Eur J Radiol 2009; 72(2): 202–208. doi: 10.1016/j.ejrad.2009.05.060.
41. Válek V, Zbořil V, Hep A et al. Tenké střevo – radiologická diagnostika patologických stavů. 1. vyd. Brno: NCONZO 2003: 61–68.
42. Jaffe TA, Miller CM, Merkle EM. Practice patterns in imaging of the pregnant patient with abdominal pain: a survey of academic centers. AJR Am J Roentgenol 2007; 189(5): 1128–1134. doi: 10.2214/AJR.07.2277.
43. Patenaude Y, Pugash D, Lim K et al. Diagnostic Imaging Committee. Society of Obstetricians and Gynaecologists of Canada. The use of magnetic resonance imaging in the obstetric patient. J Obstet Gynaecol Can 2014; 36(4): 349–363. doi: 10.1016/s1701-2163(15)30612-5.
44. Ray JG, Vermeulen MJ, Bharatha A et al. Association between MRI exposure during pregnancy and fetal and childhood outcomes. JAMA 2016; 316(9): 952–961. doi: 10.1001/jama.2016.12126.
45. Ilnyckyji A, Blanchard JF, Rawsthorne P et al. Perianal Crohn’s disease and pregnancy: role of the mode of delivery. Am J Gastroenterol 1999; 94(11): 3274–3278. doi: 10.1111/j.1572- 0241.1999.01537.x.
46. Brandt LJ, Estabrook SG, Reinus JF. Results of a survey to evaluate whether vaginal delivery and episiotomy lead to perineal involvement in women with Crohn’s disease. Am J Gastroenterol 1995; 90(11): 1918–1922.
47. Beniada A, Benoist G, Maurel J et al. Inflammatory bowel disease and pregnancy: report of 76 cases and review of the literature. J Gynecol Obstet Biol Reprod (Paris) 2005; 34(6): 581–588. doi: 10.1016/s0368-2315(05)82883-2.
48. van Assche G, Dignass A, Reinisch W et al. The second European evidence-based consensus on the diagnosis and management of Crohn’s disease: special situations. J Crohns Colitis 2010; 4(1): 63–101. doi: 10.1016/j.crohns.2009.09.009.
49. Kitayama T, Funayama Y, Fukushima K et al. Anal function during pregnancy and postpartum after ileal pouch anal anastomosis for ulcerative colitis. Surg Today 2005; 35(3): 211–215. doi: 10.1007/s00595-004-2926-y.
50. Remzi FH, Gorgun E, Bast J et al. Vaginal delivery after ileal pouch-anal anastomosis: a word of caution. Dis Colon Rectum 2005; 48(9): 1691–1699. doi: 10.1007/s10350-005-0124-7.
51. Hahnloser D, Pemberton JH, Wolff BG et al. Pregnancy and delivery before and after ilealpouch-anal anastomosis for inflammatory bowel disease: immediate and long-term consequences and outcomes. Dis Colon Rectum 2004; 47(7): 1127–1135. doi: 10.1007/s10350-004- 0569-0.
52. Lepistö A, Sarna S, Tiitinen A et al. Female fertility and childbirth after ileal pouch-anal anastomosis for ulcerative colitis. Br J Surg 2007; 94(4): 478–482. doi: 10.1002/bjs.5509.
53. Hill J, Clark A, Scott NA. Surgical treatment of acute manifestations of Crohn’s disease during pregnancy. J R Soc Med 1997; 90(2): 64–66. doi: 10.1177/014107689709000203.
54. Adamina M, Bonovas S, Raine T et al. ECCO guidelines on therapeutics in Crohn’s disease: surgical treatment. J Crohns Colitis 2020; 14(2): 155–168. doi: 10.1093/ecco-jcc/jjz187.
55. Kizer NT, Powell MA. Surgery in the pregnant patient. Clin Obstet Gynecol 2011; 54(4): 633–641. doi: 10.1097/GRF.0b013e318236eb0d.
56. Germain A, Brunaud L. Visceral surgery and pregnancy. J Visc Surg 2010; 147(3): e129–e135. doi: 10.1016/j.jviscsurg.2010.07.005.
57. Pearl J, Price R, Richardson W et al. Society of American Gastrointestinal Endoscopic Surgeons. Guidelines for diagnosis, treatment, and use of laparoscopy for surgical problems during pregnancy. Surg Endosc 2011; 25(11): 3479–3492. doi: 10.1007/s00464-011-1927-3.
58. Yakasai IA, Bappa LA. Diagnosis and management of adnexal masses in pregnancy. J Surg Tech Case Rep 2012; 4(2): 79–85. doi: 10.4103/2006-8808.110249.
59. Kunovsky L, Kala Z, Mitas L et al. Rare cases imitating acute appendicitis: three case reports and a review of literature. Rozhl Chir 2017; 96(2): 82–87.
60. Kirshtein B, Perry ZH, Avinoach E et al. Safety of laparoscopic appendectomy during pregnancy. World J Surg 2009; 33(3): 475–480. doi: 10.1007/s00268-008-9890-4.
61. Corneille MG, Gallup TM, Bening T et al. The use of laparoscopic surgery in pregnancy: evaluation of safety and efficacy. Am J Surg 2010; 200(3): 363–367. doi: 10.1016/ j.amjsurg.2009.09.022.
62. Sadot E, Telem DA, Arora M et al. Laparoscopy: a safe approach to appendicitis during pregnancy. Surg Endosc 2010; 24(2): 383–389. doi: 10.1007/s00464-009-0571-7.
63. Walker HG, Al Samaraee A, Mills SJ et al. Laparoscopic appendicectomy in pregnancy: a systematic review of the published evidence. Int J Surg 2014; 12(11): 1235–1241. doi: 10.1016/j. ijsu.2014.08.406.
64. Toiyama Y, Araki T, Yoshiyama S et al. Fulminant ulcerative colitis during pregnancy successfully treated by three-stage operation. J Gastroenterol 2004; 39(3): 300–301. doi: 10.1007/s00535-003-1295-4.
65. Quddus A, Martin-Perez B, Schoonyoung H et al. Toxic megacolon during pregnancy in ulcerative colitis: a case report. Int J Surg Case Rep 2015; 11: 83–86. doi: 10.1016/j.ijscr.2015.04. 033.
66. Germain A, Chateau T, Beyer-Berjot L et al. Surgery for Crohn΄s disease in pregnancy: a nationwide survey. United European Gastroenterol J 2020; 8(6):736–740. doi: 10.1177/2050 640620921060.
67. Selinger CP, Leong RW, Lal S. Pregnancy related issues in inflammatory bowel disease: evidence base and patients’ perspective. World J Gastroenterol 2012; 18(21): 2600–2608. doi: 10.3748/wjg.v18.i21.2600.
68. Biedermann L, Rogler G, Vavricka SR et al. Pregnancy and breastfeeding in inflammatory bowel disease. Digestion 2012; 86(Suppl 1): 45– 54. doi: 10.1159/000341941.
69. Šerclová Z, Ryska O, Bortlík M et al. Doporučené postupy chirurgické léčby pacientů s idiopatickými střevními záněty – 2. část: Crohnova nemoc. Gastroent Hepatol. 2015; 69(3): 223–238. doi: 10.14735/amgh2015223.
70. Kunovsky L, Marek F, Kala Z et al. Possibilities of minimally invasive surgery in patients with Crohn’s disease and ulcerative colitis. Gastroent Hepatol 2017; 71(1): 29–35. doi: 10.14735/amgh201729.
71. Marek F, Kunovsky L, Grolich T et al. Acute abdomen in patients with Crohn’s disease case reports. Rozhl Chir 2019; 98(1): 31–34.
72. Visser BC, Glasgow RE, Mulvihill KK et al. Safety and timing of nonobstetric abdominal surgery in pregnancy. Dig Surg 2001; 18(5): 409–417. doi: 10.1159/000050183.
73. Kunovsky L, Hemmelova B, Kala Z et al. Crohn disease and pregnancy: a case report of an acute abdomen. Int J Colorectal Dis 2016; 31(8): 1493–1494. doi: 10.1007/s00384-016-2554-1.
74. Porter H, Seeho S. Obstructed ileostomy in the third trimester of pregnancy due to compression from the gravid uterus: diagnosis and management. BMJ Case Rep 2014; 2014: bcr2014205884. doi: 10.1136/bcr-2014-205884.
75. Spring A, Lee M, Patchett S et al. Ileostomy obstruction in the third trimester of pregnancy. Colorectal Dis 2012; 14(9): e631–e632. doi: 10.1111/j.1463-1318.2012.02972.x.
76. Bemelman WA, Warusavitarne J, Sampietro GM et al. ECCO-ESCP consensus on surgery for Crohn΄s disease. J Crohns Colitis 2018; 12(1): 1–16. doi: 10.1093/ecco-jcc/jjx061.
77. Rogler G. Chronic ulcerative colitis and colorectal cancer. Cancer Lett 2014; 345(2): 235–241. doi: 10.1016/j.canlet.2013.07.032.
78. Ross H, Steele SR, Varma M et al. Practice parameters for the surgical treatment of ulcerative colitis. Dis Colon Rectum 2014; 57(1): 5–22. doi: 10.1097/DCR.0000000000000030.
79. Šerclová Z, Ryska O, Bortlík M et al. Doporučené postupy chirurgické léčby pacientů s idiopatickými střevními záněty – 3. část: ulcerózní kolitida. Gastroent Hepatol 2016; 70(3): 252–261. doi: 10.14735/amgh2015252.
80. Teeuwen PH, Stommel MW, Bremers AJ et al. Colectomy in patients with acute colitis: a systematic review. J Gastrointest Surg 2009; 13(4): 676–686. doi: 10.1007/s11605-008-0792-4.